|
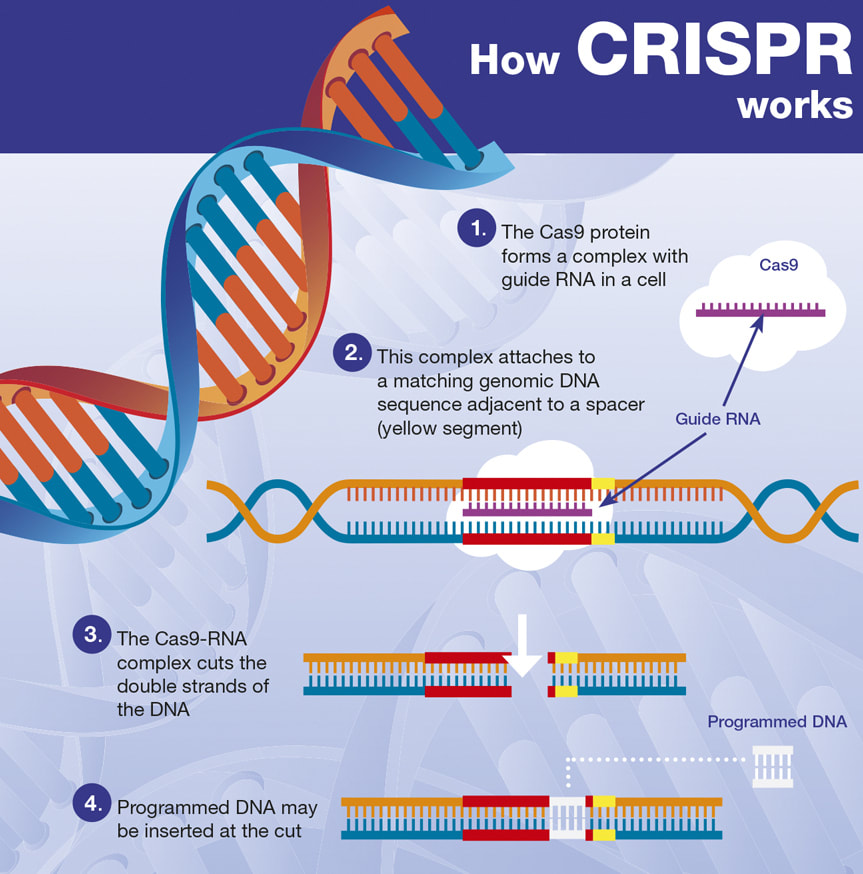
CRISPR is an acronym for ‘clustered regularly interspaced short palindromic repeats,’ which are nucleotide sequences and spacers found in specialized regions of DNA from prokaryotic organisms (usually single-celled bacteria or archaea that lack a membrane-bound nucleus and membrane-bound organelles) [1]. These regions are accompanied by Cas proteins and together they make up antiviral utilities which detect and degrade exogenous DNA and RNA from infiltrating viruses (there is also evidence of CRISPR-Cas systems helping with endogenous transcriptional control) [2]. CRISPR sequences originate in prokaryotes from past viral infections or plasmid exposures, and Cas (CRISPR-associated) proteins are enzymes that use CRISPR sequences to specifically locate and cleave matching or complimentary DNA fragments to thwart subsequent infection or unwanted genome alterations through the uptake of foreign nucleic acids [3] [4].
The CRISPR-Cas system can be hijacked or differently employed as a gene therapy tool, and this fact has led to the development of a gene editing technology named CRISPR-Cas9 (in which one particular Cas protein, Cas9, is utilized) [5]. CRISPR-Cas9 allows for targeted DNA deletions, insertions, and substitutions, and thus can be compared to a word processor’s cut and paste function. Viral means and non-viral means (like lipid- or polymer-based nanocarriers) for delivering the CRISPR-Cas9 vehicle to target cells currently exist (note that viral vectors carry a risk of cancer induction and violent immune responses) [6]. And a related technology involving nuclease-deactivated Cas9 (CRISPR-dCas9), can be used to epigenetically alter the activity of a particular gene without the normal DNA-cutting action of Cas9 [7]. CRISPR-Cas9 is not a perfect instrument however, as there can be variable accuracy and efficiency issues. And because gene editing literally consists of making changes to an organism’s genetic code, there are major ethical concerns with its extrapolated use (beyond simply correcting genetic defects in those with an inherited genetic disease and into extensions like germline engineering) [8]. Now, Duchenne muscular dystrophy (DMD) stands as an inherited genetic disease in which mutations in the DMD gene typically cause a failure to manufacture the protein dystrophin, an integral component of the dystrophin-glycoprotein complex found in heart and skeletal muscle cells that serves as a cytoskeleton-extracellular matrix bridge [9]. Without the mechanical stabilization and signaling roles of the dystrophin-glycoprotein complex, cardiac and skeletal myocytes degenerate with use and progressive muscle wasting results [10]. DMD patients typically experience cardiac or respiratory failure during the third or fourth decade of life resulting in premature death [11]. The correction of DMD mutations via exon skipping or exon deletion with the use of CRISPR-Cas9 can restore expression of the dystrophin protein and rescue the function of cardiac and skeletal muscle cells, and this has been safely demonstrated in mouse models of Duchenne muscular dystrophy [12] [13] [14]. Other strategies for remedying DMD mutations with the help of CRISPR-Cas9 are available too [15]. DMD mutations have also been corrected using CRISPR-Cas9 in human cells in vitro [16] [17] [18] [19] [20]. And dystrophin expression and improved muscle histology have been seen in a canine model of DMD [21]. While thousands of DMD mutations have been identified in patients with Duchenne muscular dystrophy, CRISPR technologies have exhibited efficacy in restoring dystrophin expression in human cells, and thus offer great promise for an otherwise incurable disease [22]. In conclusion, even though CRISPR systems remain highly advantageous because they are capable of permanently fixing genetic defects in those with DMD, there is an unpredictability to gene editing, and undesired mutations, off-target effects, and dangerous immune responses can be seen [23]. Genetic mosaicism has helped teach us that the human genome is much more malleable than previously believed, which carries with it both good and bad potentials [24]. Gene editing has the capacity to powerfully treat and even rectify many inherited genetic diseases, but certainly we must be careful to not get lured into playing God. Kept within strict boundaries, gene therapy can continue to grow into a medical revolution, and very soon, CRISPR systems may become easily accessible to those who truly need them. References:
0 Comments
Your comment will be posted after it is approved.
Leave a Reply. |
AuthorDenton Coleman is an Exercise Physiologist and Medical Researcher. Archives
October 2023
Categories |

 RSS Feed
RSS Feed
